OEM Services
Presently Seeking OEM Customers.
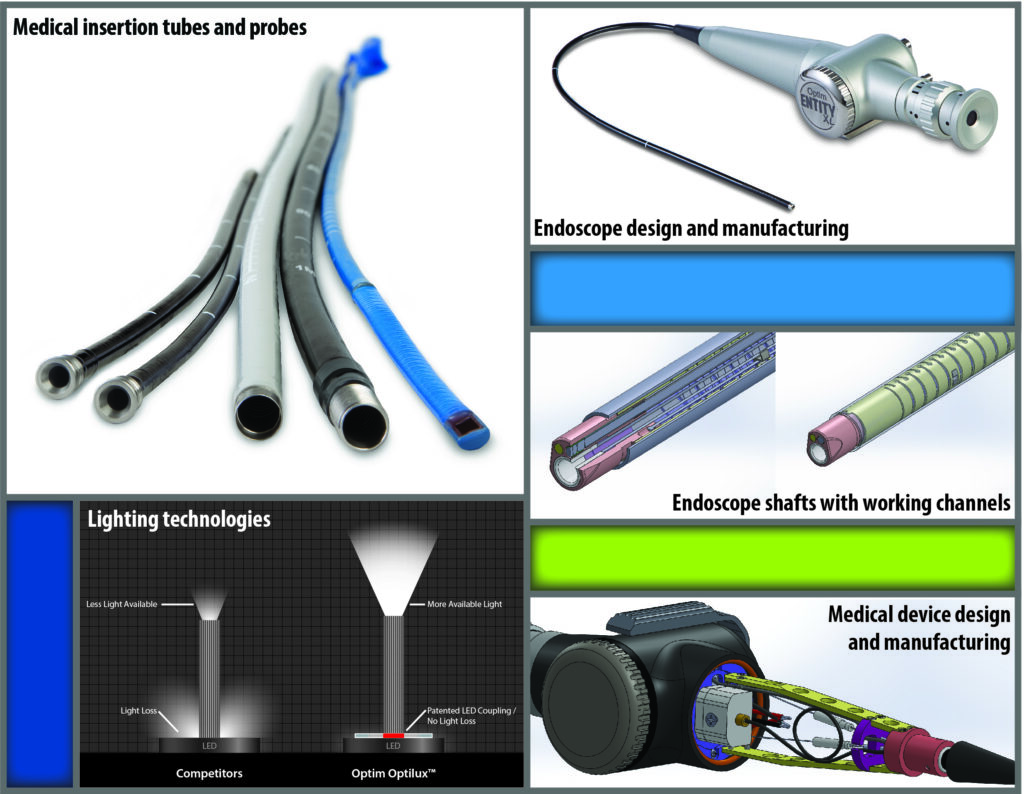
Optim offers design and manufacturing expertise in fiber optic endoscopes, videoscopes, endoscope sub-assemblies and components for medical and industrial imaging applications. Leveraging our development and manufacturing experience in prototype production, product validation to final product release allows your company to focus on design requirements, product launch, marketing, and distribution.
Optim’s OEM customers include large medical device companies, endoscope service companies, early-stage companies and distributors. These customers rely on our knowledge of fiber optic imaging and insertion
tube manufacturing to conceive and build products that meet their unique requirements. Project and product confidentiality is assured under Non-Disclosure Agreements.
Optim is an FDA registered manufacturing facility to FDA 21CFR Part 820 and complies with the MDD 93/42/EEC Annex VII through our authorized representative, Scanlan Group B.V.






